Lecture - 7 ( temperature and thermometer )
Temperature
Temperature is a parameter which represents average random energy per unit molecule. As random energy per unit molecules decreases then it means that temperature will get reduce and vice-versa.
For more details on temperature or difference between temperature and heat check out Lecture - 6.
Conversion of celsius to fahrenheit scale
There there is no Rocket Science behind it you just need to remember a simple formula for this conversion and that is-
Tc / 100 = (Tf - 32) / 180
'Tc ' represents temperature in degree Celsius scale
'Tf 'represents temperature in fahrenheit scale
Celsius and Fahrenheit scale are based upon 2 reference points which are boiling and freezing. In Celsius scale 0 denotes the freezing point of water and 100 denotes the boiling point of water. Whereas in fahrenheit scale reading of 32 denotes freezing point of water and reading of 212 denotes boiling point of water.
Conversion of celsius to kelvin scale
Formula for this is even more simple and that is-
Tk = Tc + 273.15
'Tk ' represents temperature in kelvin
Kelvin scale is absolute thermodynamic temperature scale because it is single point Reference scale. The reference point selected for Kelvin scale is triple point of water. According to internationally accepted definition, 1 kelvin is defined as 1/273.16 of triple point of water.
Zero Kelvin temperature or -273.15 degree celsius temperature is minimum possible temperature but in theories, practically 0 Kelvin temperature is impossible to achieve and for understanding the concept behind this you can check Intresting facts.
Conversion of celsius to fahrenheit scale
There there is no Rocket Science behind it you just need to remember a simple formula for this conversion and that is-
Tc / 100 = (Tf - 32) / 180
'Tc ' represents temperature in degree Celsius scale
'Tf 'represents temperature in fahrenheit scale
Celsius and Fahrenheit scale are based upon 2 reference points which are boiling and freezing. In Celsius scale 0 denotes the freezing point of water and 100 denotes the boiling point of water. Whereas in fahrenheit scale reading of 32 denotes freezing point of water and reading of 212 denotes boiling point of water.
Conversion of celsius to kelvin scale
Formula for this is even more simple and that is-
Tk = Tc + 273.15
'Tk ' represents temperature in kelvin
Kelvin scale is absolute thermodynamic temperature scale because it is single point Reference scale. The reference point selected for Kelvin scale is triple point of water. According to internationally accepted definition, 1 kelvin is defined as 1/273.16 of triple point of water.
Zero Kelvin temperature or -273.15 degree celsius temperature is minimum possible temperature but in theories, practically 0 Kelvin temperature is impossible to achieve and for understanding the concept behind this you can check Intresting facts.
Thermometers
thermometer are based on the principle of finding the thermometric property. Thermometric property is a property which helps in finding temperature. For example in case of a regular thermometer change in volume of Mercury shows us the reading of temperature, here in this case volume is our thermometric property.
Types of thermometers
Thermistor or resistance thermometer
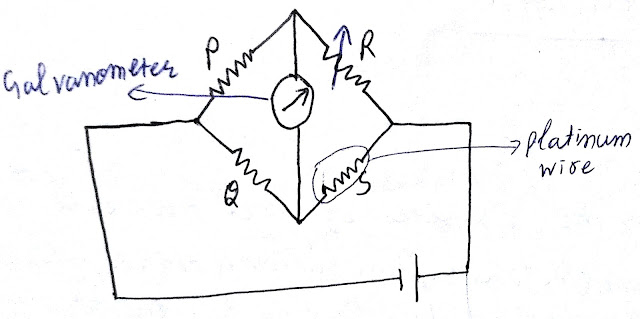
This thermometer works on the principle of wheatstone bridge, here thermometric property is resistance.
Let me explain the working of this thermometer with the help of an example. Consider the below figure in which four resistance are shown that is P, Q, R and S. Here value of P and Q resistance are known to us, say P = 5 ohm and Q = 10 ohm. R is our variable resistance and its value will change with the changes in the reading of galvanometer. Let for zero deviation of galvanometer resistance R comes to be 8 ohm. Value of resistance S depends upon material to material and this resistance will help us to find out the temperature. Here I am considering a Platinum wire.
According to wheatstone Bridge principle we know
P/Q = R/S
5/10 = 8/S
S = 16 ohm
To find out temperature from this resistance just put the value of resistance in below equation-
Let me explain the working of this thermometer with the help of an example. Consider the below figure in which four resistance are shown that is P, Q, R and S. Here value of P and Q resistance are known to us, say P = 5 ohm and Q = 10 ohm. R is our variable resistance and its value will change with the changes in the reading of galvanometer. Let for zero deviation of galvanometer resistance R comes to be 8 ohm. Value of resistance S depends upon material to material and this resistance will help us to find out the temperature. Here I am considering a Platinum wire.
According to wheatstone Bridge principle we know
P/Q = R/S
5/10 = 8/S
S = 16 ohm
To find out temperature from this resistance just put the value of resistance in below equation-
S = So (1 + αt + βt^2 )
Here So , α and β are constants and t represents the temperature. Just put the value of S in this equation and we get temperature reading.
Thermocouple
Thermocouple is based on seebeck effect. According to this effect if two different metals are joined together to form two different junctions and the junctions are maintained at different temperature, then EMF is produced which is directly proportional to the temperature difference. by measuring the EMF with the help of voltmeter the unknown temperature can be formed. Circuit diagram for this is shown below.
So from this phenomenon one more effect comes into picture and it's name is peltier effect. Peltier effect is the opposite of the seebeck effect and is used for thermoelectric refrigeration. Here potential difference is developed between two different metals due to which temperature of one metal increase and other metal decrease. Circuit diagram of peltier effect is shown below.
Constant volume Gas thermometer
To use this thermometer gas should be ideal or perfect gas. Here volume will remain constant so when temperature increases energy associated per unit molecule increases and molecules striking the wall increases but our volume is constant so pressure will increase and the reading of increase in pressure helps us to find out the temperature. With the help of ideal gas equation change in pressure will give the reading of temperature. Here pressure is thermometric property.
PV = mRT
P/T = constant
P1 / T1 = P2/T2
We already know initial pressure and temperature and reading will show us final pressure. Solve the equation and we get T2 which is our required temperature.
Constant pressure Gas thermometer
This also works on ideal or perfect gas. In this thermometer as the temperature changes energy associated with per unit molecule increases due to which collision on the wall of cylinder and Piston increases, but our piston is free to move so pressure will remain constant and volume of the Piston cylinder arrangement changes, and this change in volume gives us the reading of temperature. Here volume is thermometric property.
PV = mRT
V/T = constant
V1 / T1 = V2 / T2
T2 is required temperature
constant volume Gas thermometer and constant pressure Gas thermometer both are also called as ideal Gas thermometer because material of construction is ideal gas. These thermometers are independent of material of construction that is if the gas is an ideal gas it doesn't matter which gas it is, result will remain same.







Comments
Post a Comment